This course is for early-stage Medical Device, MedTech and HealthTech organisations planning to provide medical devices and related services that consistently meet customer and applicable regulatory requirements of ISO 13485. Your organisation may be involved in one or more stages of the life-cycle such as design and development, production, storage and distribution, installation, or servicing of a medical device and design and development or provision of associated activities (e.g. technical support). ISO 13485 can also be used by suppliers that provide product and/or services to such organisations.
Participants will gain knowledge and learn process steps to facilitate the implementation of an effective QMS aligned with or certified to ISO 13486:2016.
Applying ISO 13485 helps ensure that customers get consistent, good-quality products and services, which in turn brings many business benefits and commercial success.
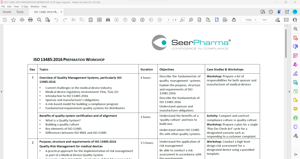
Content
- Overview of Quality Management Systems, particularly ISO 13485:2016
- Benefits of quality system certification and of alignment
- Purpose, structure and requirements of ISO 13485:2016
- Quality Risk Management for medical devices (ISO 14971)
- Management Responsibility
- Resource Management
- Product realization
- Measurement, Analysis and Improvement
- Overview of application process for ISO 13485:2016
- Development of documentation required for ISO 13485:2016
- Preparation for applying for ISO 13485:2016
Click here for a full course outline (PDF)
Participants
This course is open to anyone interested in learning more about the application of ISO 13485:2016 requirements and benefits of a quality management system for medical devices. You may be a subject matter expert, researcher, start-up, supplier or a graduate with interest.
Limited to 25 participants maximum.
Format and Course Length
Delivery will be a mix of theory and interactive case studies and workshops to accelerate your learning with practical skills. Delivery will run over 4 days (Monday to Thursday) with group participation exercises and periodic breaks to keep you engaged.
What Do I Need?
You just need to attend with a positive attitude and willingness to participate. SeerPharma will provide you with the training materials and catering (food and beverage) will be provided each day.
Cost
With the support of industry partners to subsidise this training, you only need to pay a small co-contribution of $440 (including GST) to attend this 4-day workshop. Upfront payment will be required to participate. On-site parking at additional cost.
Session and Registration
| Dates | Venue |
| To be advised |
To be advised |
If you can't see the booking window to select tickets below, please click here.
Contact us to discuss your interest in a public or private delivery of this course.